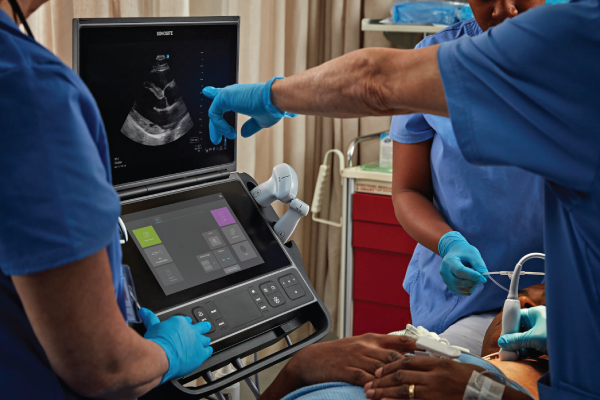
Cost of an Ultrasound System
Understanding the Cost of Owning an Ultrasound SystemPoint-of-care ultrasound (POCUS) has become an essential tool in modern medicine, but how much does owning one really cost? When considering POCUS for your practise, understanding the full picture of costs involved—from initial purchase to system maintenance—is crucial. …
#PractiseMakesPOCUS
Clinicians often struggle with the complexities of mastering POCUS scanning techniques. It's not the tech they struggle with, it's the scanning. Discover Sonosite Scan Along - available on all our newest FUJIFILM Sonosite systems. …
Did You Know that Sonosite has a New Vascular Access Education Partner?

Did You Know that FUJIFILM Sonosite has a new partner to provide vascular access education to our customers? With education being one of our core pillars, we strive to ensure that your department and clinicians are adequately equipped with the knowledge and training to administer care to patients using point-of-care ultrasound. …
Designed for Durability: The Rigorous Testing Process of Sonosite Transducers

Introduction: Setting the Bar HighIn Point-of-Care-Ultrasound, the durability of equipment is paramount. Without quality systems that are ready to use, the immediacy of patient care can be negatively impacted. To ensure reliability and up-time, Sonosite transducers are tested to the limit. This process consists of extensive testing protocols that go beyond industry norms. …
Empowering Healthcare Providers with Sonosite's Voice Assist
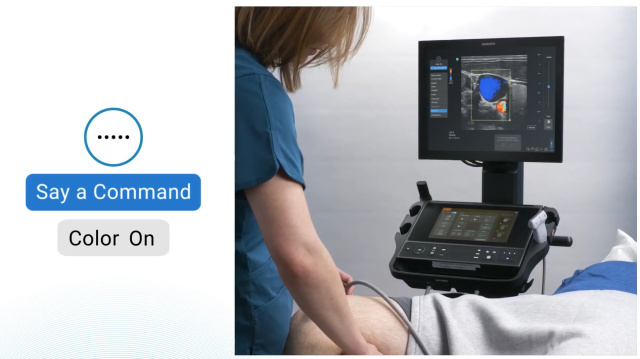
In today's healthcare landscape, institutions face a daunting challenge: maintaining high-quality patient care, often with limited staffing.
…
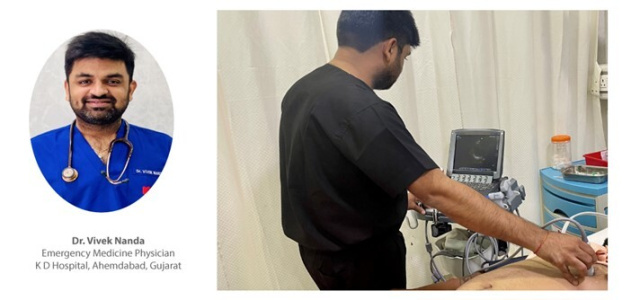
POCUS Profile: Dr. Tushar B Munnoli
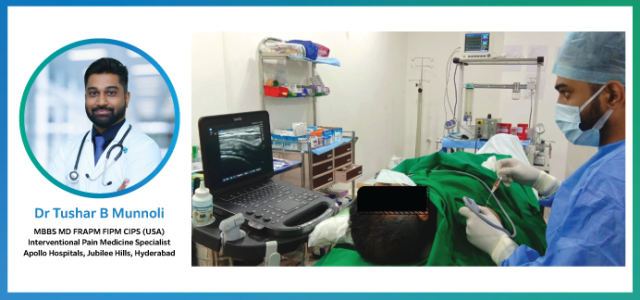
We recently interacted with Dr. Tushar B. Munnoli who is a renowned Interventional Pain Medicine specialist currently practicing at Apollo Hospitals, Jubilee Hills, Hyderabad. After his post-graduation in Anesthesia, Dr. Munnoli pursued a postdoctoral fellowship in Regional Anesthesia and Acute Pain Management. He holds a diploma in Musculoskeletal ultrasound and is a certified Interventional Pain Sonologist (CIPS) from the World Institute of Pain (USA). …
Joining the Mission with the Friends of Fiji Heart Foundation

The Mission
Since the formation of the Friends of Fiji Heart Foundation (FOFHF) in 2006, the Foundation has sent a mission to Fiji for each successive year to provide FREE cardiac surgery to the underprivileged people of Fiji. Over the years the Foundation’s focus has expanded to include open heart surgery, pacemaker checks and implants, angiography, stenting, etc. Since 2006, treatments worth over NZ$10m have been performed with ZERO administrative costs. …
Ryan Hebbler

FUJIFIILM Sonosite employees are passionate about many things, helping people is at the top of the list. As is the case for Ryan Hebbler, our Director of Strategic Accounts, which means helping customers, colleagues, clinicians, and everyone else he deals with. He's passionate about ultrasound, and it shows. …
Carena Holmes

Carena Holmes, senior systems engineer sees the big picture and the details. She is always thinking about how to improve our systems and how teams can work more harmoniously. When asked about her favourite project, she replies with a secretive smile, but that just goes to show how interesting the work she's doing is. She's a creative thinker and a passionate advocate for her work. …
Keith Williams

Keith Williams, Executive Director of Programme Management at FUJIFILM Sonosite, is celebrating his 25th anniversary with the company. When asked what keeps him coming back, Williams said, "It's the people. I get to work with some phenomenally talented and passionate people who are dedicated to making a difference in the world. And the products we make have a really important role to play in that." In addition to his work at FUJIFILM Sonosite, Williams is also a passionate music fan. He shared his top 5 songs of all time with, which you can enjoy below. …
From the CEO: Find Work that Matters

Find Work that Matters at FUJIFILM SonositeLife is finite, which makes time our most precious commodity.The COVID pandemic has taken this concept and dropped it right in the lap of millions of working people in this country. “Why am I spending the majority of my waking hours doing this work, and is that how I want to spend the time I have left on this planet?” Welcome to the Great Resignation. …
From the CEO: Core Values

Core Values: A focus on HOW we face challenges together
The last few years have challenged us all. They’ve made us think deeply about what matters in our lives. Many have been faced with choices they never dreamed they’d have to make, especially in the early days of the pandemic. Do I work and risk illness or possibly death, or expose my loved ones to the same? Do I stop working and possibly lose my home? If you own or run a business, do you remain open in an effort to keep your team employed or do you shut down to reduce the spread of disease? …
Top 10 Perioperative Applications of Point-of-Care Ultrasound for Anaesthesiologists
OR Today
Davinder S. Ramsingh, MD
With the release on May 30, 2019 of the final report from the Federal government’s Pain Management Best Practises Inter-Agency Task Force, anaesthesiologists need specific case studies on how to implement recommendations in their hospitals. This OR Today article, published by Davinder S. Ramsingh, MD, Director of Research and Perioperative Ultrasound at Loma Linda University Medical Centre, Department of Anaesthesiology, shares ten applications of point-of-care ultrasound, a modality mentioned in Perioperative Surgical Home (PSH) and Enhanced Recovery After Surgery (ERAS)
clinical practise guidelines. The Task Force’s final report recommends to clinicians the use of multidisciplinary and multimodal approaches for perioperative pain control in selected patients at higher risk for opioid use disorder. Read Dr. Ramsingh’s article to learn more about how anaesthesiologists and pain medicine specialties can incorporate point-of-care ultrasound with regional anaesthesia or TEE, both potential elements of these multi-modality approaches.
…